Crown Lengthening - Concord, NH
Achieve an Even, Healthy Smile With Specialized Crown Lengthening Services
Schedule ConsultationCrown Lengthening Procedures Deliver Exceptional End Results
At times, excess gum tissue creates both cosmetic and functional challenges in the mouth. Conditions like a “gummy smile” or tooth decay deep below the gum line require the removal of gum and bone tissue to promote better oral health and cosmetic results. Excess gum tissue causes teeth to appear too short. It also provides additional spaces for disease-causing bacteria to hide. At Cornerstone Periodontics, we provide both cosmetic and functional crown lengthening. These specialized services restore health and an even appearance to your smile when tooth surface exposure is necessary.
How Do Advanced Crown Lengthening Procedures Work?

Esthetic Crown Lengthening
This periodontal plastic surgery procedure sculpts and reshapes the gum line for a more cosmetic, uniform smile. We gently remove excess gum and sometimes even bone tissue to reveal more surface area of your teeth. A better gum-to-teeth ratio is easily achieved with a simple esthetic crown lengthening procedure.
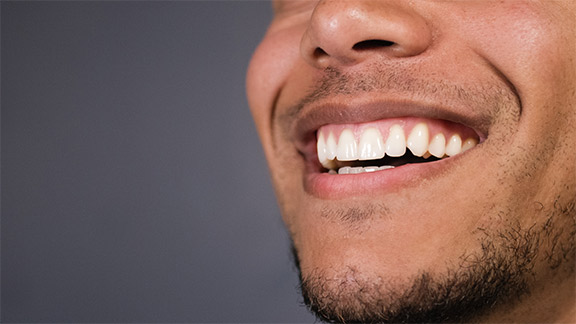
Functional Crown Lengthening
Deep decay below the gum line is often difficult to access. In such cases, your general dentist may refer you to our practice for functional crown lengthening. We reduce the height of the gum tissue and bone to enable access to areas of concern. Your dentist is then able to treat your tooth and place a properly fitting restoration.
Look Forward to the Benefits Crown Lengthening Brings to Your Smile
Both esthetic and functional crown lengthening bring exceptional cosmetic and clinical results to your smile. Benefits of these common procedures include:
- Healthier gums
- Even, balanced gum line
- Better homecare ability
- Reduced risk of gum disease
- Easier access to tooth decay or damage
- Appealing cosmetic appearance
- Boost in self-confidence
- Proper spacing for restorative needs
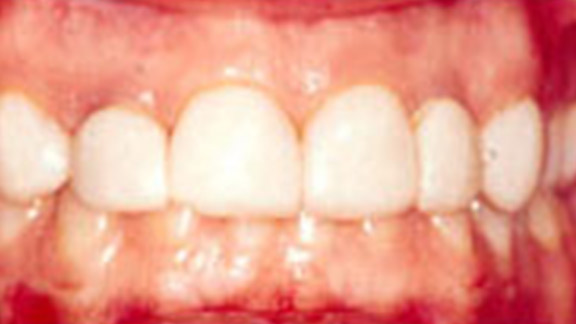
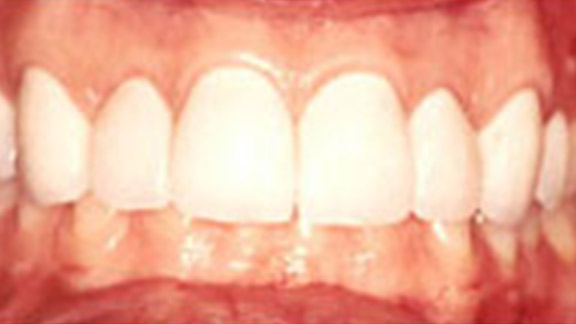
Crown Lengthening Case 1
